Rice eyes risks of quantum dots
Researchers test nanocrystals for health, environmental concerns
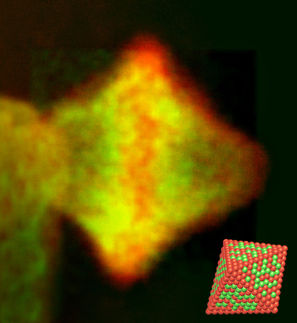
quantum dots have the potential to bring many good things into the world: efficient solar power, targeted gene and drug delivery, solid-state lighting and advances in biomedical imaging among them. But they may pose hazards as well. A team of rice researchers has been working to discover the health risks of quantum dots, molecule-sized semiconducting nanocrystals that are generally composed of heavy metals surrounded by an organic shell.
Pedro Alvarez, Rice's George R. Brown Professor and chair of the Civil and Environmental Engineering Department, published a paper in ACS Environmental Science and Technology showing that under even mildly acidic or alkaline conditions, the shells can break down, releasing their toxic contents into the body or the environment. He coauthored the paper with colleagues Vicki Colvin, the Pitzer-Schlumberger Professor of Chemistry and in chemical and biological engineering, researcher Shaily Mahendra and postdoc Huiguang Zhu.
"We're interested in the long-term implications of nanotechnology, and we recognized quantum dots are going to be produced in large quantities," said Mahendra, who did the bulk of the research. "We thought we should be proactive in studying their effects so that we can take part in the development of safety guidelines."
The dots, 1/50,000ths the width of a human hair, were found to be safe in applications with a neutral pH environment. However, the study suggested when such products are discarded, they can eventually release their toxins into the environment.
"In that way, quantum dots resemble batteries," said Alvarez, referring to common nickel-cadmium cells that people are warned not to throw in the trash. "They're often made of coatings that are biocompatible and stable in water, but the moment we lose that coating, which can happen through a variety of mechanisms, they can release toxic compounds."
Used in solar cells, quantum dots may be quickly weathered by acid rain, he said. Another concern is that acids in the body could break down dots used in such medical applications as in vivo imaging. "If they degrade faster than they can be excreted, there's the potential for heavy metals to be released into the body," said Alvarez. "Then their impact becomes a question of dose."
The team tested its theories on common bacteria that serve as models of cell toxicity and indicators of environmental health. At near-neutral pH, bacteria exposed to quantum dots containing cadmium and selenium showed decreased rates of growth but did not die. But in moderately acidic or alkaline conditions, quantum dot shells decomposed more rapidly, killing the bacteria in a matter of hours.
On the positive side, the study also found certain proteins and such natural organic matter as humic acids may mitigate the effects of decomposing quantum dots by coating them or by complexing the metal ions released, making them less toxic.
The researchers cautioned that short-term studies can't easily predict whether toxins released by quantum dots will build up in the body over time. "We hope our work will stimulate research by other labs into the release dynamics," said Alvarez.
Most read news
Topics
Organizations
Other news from the department science
These products might interest you

Dursan by SilcoTek
Innovative coating revolutionizes LC analysis
Stainless steel components with the performance of PEEK - inert, robust and cost-effective

OCA 200 by DataPhysics
Using contact angle meter to comprehensively characterise wetting behaviour, solids, and liquids
With its intuitive software and as a modular system, the OCA 200 answers to all customers’ needs

Tailor-made products for specific applications by IPC Process Center
Granulates and pellets - we develop and manufacture the perfect solution for you
Agglomeration of powders, pelletising of powders and fluids, coating with melts and polymers

Get the chemical industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.