New X-ray method allows scientists to probe molecular explosions
Summer blockbuster season is upon us, which means plenty of fast-paced films with lots of action. However, these aren't new releases from Hollywood studios; they're one type of new "movies" of atomic-level explosions that can give scientists new information about how X-rays interact with molecules.
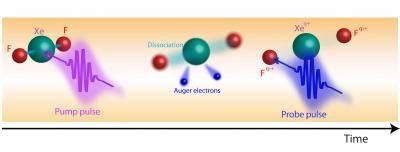
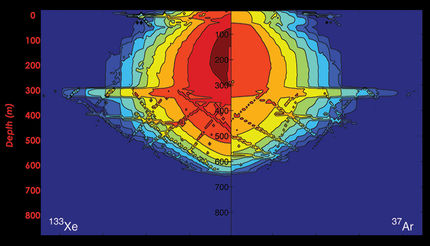
A team led by researchers from the Argonne National Laboratory used the high-intensity, quick-burst X-rays provided by the Linac Coherent Light Source (LCLS) at SLAC National Accelerator Laboratory to look at how the atoms in a molecule change when the molecule is bombarded with X-rays. This schematic shows the dissociation of a xenon difluoride molecule during the X-ray pump/X-ray probe process.
Antonio Picon
A team led by researchers from the U.S. Department of Energy's Argonne National Laboratory used the high-intensity, quick-burst X-rays provided by the Linac Coherent Light Source (LCLS) at SLAC National Accelerator Laboratory to look at how the atoms in a molecule change when the molecule is bombarded with X-rays.
"The LCLS gives us a unique perspective on molecular dynamics because of the extremely brief X-ray pulses that we can use," said Antonio Picon, an Argonne X-ray scientist and lead author. "We're able to see how charge and energy can flow through a system with amazing precision."
By using a new method called X-ray pump/X-ray probe, the researchers were able to excite a specifically targeted inner-shell electron in a xenon atom bonded to two fluorine atoms. After the electron was excited out of its shell, the unbalanced positive charge in the rest of the molecule caused the molecule to spontaneously dissociate in a process known as "Coulomb explosion."
"The new X-ray pump/X-ray probe technique is so powerful because it allows us to shake the molecule at one point, and look at how it changes at a second point," said Argonne X-ray scientist and study author Christoph Bostedt.
The xenon difluoride molecule is only a first step for the technique. In the future, the same X-ray pump/X-ray probe method could find a broad range of applications, such as following the ultrafast structural changes that occur in light-sensitive molecules or the flow of energy in molecules. By understanding intramolecular energy flow, researchers can better develop novel materials to harness the sun's energy, such as photovoltaics and photocatalysts.
The new technique could also help researchers address challenges relating to the protein structure determination. For pharmaceutical studies, X-rays are often used to figure out the structures of proteins, but during that process they can also damage parts of them.
"This technique lets you see how neighboring atoms are affected when certain regions interact with X-rays," said Stephen Southworth, an Argonne senior X-ray scientist.
By using an X-ray pump to excite one of the innermost electrons in the molecule, the researchers were able to target one of the electrons that is most central to and characteristic of the molecule. "This technique gives us the ability to take a series of quick snapshots to see what happens when we change a fundamental part of a molecule, and what we learn from it can inform how we approach the interactions between light and molecules in the future," said Picon.
The research, which was funded by the DOE Office of Science, involved a collaboration between Argonne, SLAC, and Kansas State University. "For these kinds of studies, you really need a team that combines world leaders in X-ray sources, particle detection and sample manipulation," Southworth said.
Original publication
Other news from the department science

Get the chemical industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
Most read news
More news from our other portals
Last viewed contents

Metallic five-membered ring pushes the boundaries of aromaticity - Researchers succeed in synthesizing a molecule that should not actually exist
Building up antioxidants
Glucose_transporter
Monoamine_oxidase