Transformations of diazo compounds catalyzed by environmentally benign iron complexes
catalysis can be used to enhance the reactivity and selectivity of specific chemical transformations and decrease the amount of energy consumed by these processes. Most catalytic reactions fulfill the criteria for a sustainable transformation on the macroscopic scale, and a wide variety of efficient catalytic systems have been developed in both industry and academia. However, most of the catalysts are based on precious metals such as palladium, rhodium, ruthenium, iridium and osmium, which are becoming increasingly expensive because of the dwindling resources. Moreover, the use of heavy metals as catalysts in the pharmaceutical and agricultural industries has limitations because of toxicity.
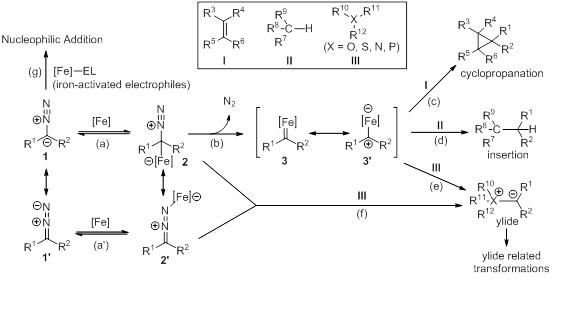
This figure shows an outline of iron-catalyzed transformations of diazo compounds.
©Science China Press
So there is an urgent need to explore effective and abundant replacements for the rare and toxic metals currently used in catalysis.
Iron is a ready available, inexpensive, and environmentally benign metal, which represents an ideal alternative to the precious metals. Iron, however, has not been developed as a catalyst to the same extent as other transition-metals with regard to use in organic transformations, especially asymmetric processes.
In a new paper scientists Shoufei Zhu and Qilin Zhou, based at Nankai University in the eastern Chinese port city of Tianjin, present an overview on recent advances in the study of iron-catalyzed diazo transformation reactions. The transition-metal-catalyzed transformations of diazo compounds are widely used in organic synthesis. The catalysts used in the reactions of diazo compounds are generally derived from rhodium, ruthenium, and copper.
Iron can undergo facile changes in its oxidation state and exhibit a distinct Lewis acid character. Iron can catalyze a variety of transformations of diazo compounds (please see Scheme 1).
For example, an iron catalyst can attack the α-carbon of diazo compound 1, and the subsequent loss of nitrogen forms iron carbene or carbenoid species 3 and 3? (processes a and b). The iron carbene or carbenoid species may undergo different carbene transfer reactions, including the cyclopropanation of olefins I (process c), C-H bond insertion of hydrocarbons II (process d), or ylide formation with heteroatom-containing compounds bearing a lone pair III (process e). The iron-activated diazo compounds 2 and 2? can also form ylides by undergoing a substitution reaction with heteroatom-containing compounds III (process f).
Furthermore, iron can act as a Lewis acid catalyst and activate electrophiles, such as imines and aldehydes, which react with diazo compound 1 (process g).
Although an iron-promoted diazo transformation was not discovered until the 1990s, it features some unique advantages. For example, the iron porphyrins are among the most efficient catalysts for the cyclopropanation reaction of olefins, which exhibit tolerance towards water, as well as several acids and bases; The iron complexes of chiral spiro bisoxazolines afford the highest level of enantioselectivity in the O-H bond insertions of alcohol and water.
Other distinct advantages that iron catalysts exhibit over other catalysts are the diazo transformations involving ylide intermediates, such as olefinantion, and the rearrangement of ammonium ylides.
These examples clearly indicate that the potential of iron-catalyzed diazo transformations is huge, although there is still a long way to go. In the future, the development of new and readily tunable iron catalysts will be the key issue in this research field.
Original publication
Shoufei Zhu, Qilin Zhou. "Iron-catalyzed transformations of diazo compound". National Science Review, (December 2014) 1 (4): 580-603.
Other news from the department science

Get the chemical industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.