New findings on ozone depletion
EU project RECONCILE: Climate change could affect recovery of the ozone layer
Good news for the ozone layer above the Arctic. The Montreal Protocol is showing effects: according to recent measurements, the ozone layer over the North Pole should recover by the end of the century. This is one of the main findings of the EU project RECONCILE, which was completed in February 2013. Scientists from Jülich and their colleagues from 35 research institutions and universities in 14 countries spent four years investigating the chemical process of ozone depletion. Their findings verified once again that chlorine compounds are indeed responsible for this. The scientists used the new insights to improve existing climate models. These models facilitate more reliable predictions on how the ozone layer will develop in future and on the possible consequences of climate change for the stratosphere.
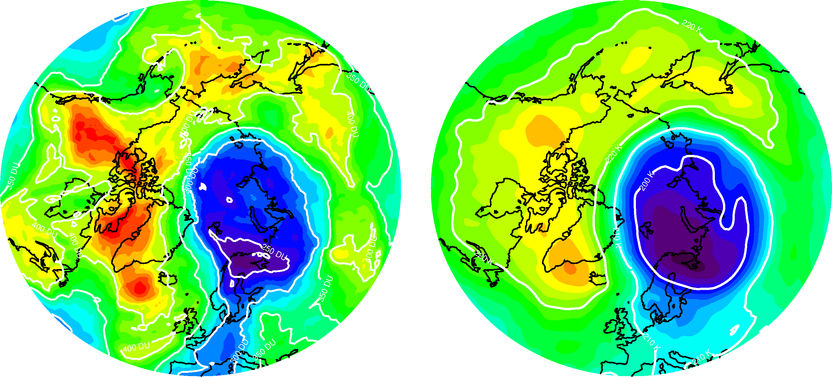
Strong ozone depletion in the cold polar vortex above the Arctic on 2 April 2011. Many of the processes that cause ozone depletion in the polar stratosphere strongly depend on temperature. This explains why climate change also affects the ozone layer. The image on the left shows the ozone layer. The purple area is where the layer is thinnest. The image on the right shows the temperature. The purple area indicates the coldest region. Source: Forschungszentrum Jülich/Marc von Hobe Ozone data: Ozone Monitoring Instrument Temperature data: European Centre for Medium-Range Weather Forecasts (ECMWF).
Forschungszentrum Jülich/ Marc von Hobe
"Even if the ozone layer recovers, climate change could alter the underlying conditions. This is yet another reason to reduce greenhouse gas emissions and stop climate change," emphasizes environmental chemist Dr. Marc von Hobe from Forschungszentrum Jülich, which coordinated RECONCILE. Climate change could alter the temperature, circulation patterns and chemical composition in the stratosphere. This also influences the ozone layer, which in turn has a bearing on temperature. Furthermore, possible ventures to mitigate climate change could have a negative impact on the ozone layer. One example is the so-called Geoengineering. The term describes the use of technological means to intervene in the geochemical or biogeochemical processes of the Earth.
In the Montreal Protocol, signed in 1987, more than 190 countries vowed to reduce the emission of chlorine containing chemicals such as chlorofluorocarbons (CFCs). A study published around six years ago indirectly called into question the role of CFCs and the Protocol. "In the project, we successfully answered some of the open questions regarding ozone loss, and we demonstrated that besides the destruction caused by chlorine, no additional chemical mechanisms play a decisive role," says Marc von Hobe. Analyses of air samples at the University of East Anglia and the University of Frankfurt as part of the project showed a clear decrease in stratospheric chlorine. Although these compounds remain in the atmosphere longer than previously thought, the scientists expect the ozone layer to recover by the end of the century.
This forecast remains unaffected by incidents such as at the beginning of 2011 when the scientists observed the most severe ozone depletion above the North Pole yet. The decisive factor two years ago was an unusually long Arctic winter. Ozone is only depleted under very cold conditions. During the polar night, the polar vortex develops – a sort of closed system with very cold air, which warmer air masses from the south cannot penetrate. Polar stratospheric clouds (PSCs) form in this vortex at temperatures below -80 °C. On their surfaces, a series of chlorine reactions is initiated that ultimately lead to ozone depletion. The longer the very cold conditions last, the more ozone is destroyed. Only when the polar vortex breaks down in spring can the ozone layer recover.
When they investigated these processes in more detail, the RECONCILE researchers unearthed a number of surprises. For example, scientists from Jülich showed that surface reactions also occur very efficiently on liquid aerosols in the air. "For chlorine chemistry, the formation of PSCs is not as important as the temperature. Theoretically, the reactions can occur anywhere where it is cold enough and sufficient chlorine is present," explains Tobias Wegner, who wrote a PhD thesis on aerosol particles and chlorine activation at Forschungszentrum Jülich. However, PSCs still play an important role in ozone depletion. Nitrogen compounds, which react with chlorine compounds to stop the ozone depletion process at the end of the winter, are bound in PSC particles and fall downwards due to gravity. Therefore, chlorine reactions can continue right into spring. "The findings of the project have completely altered our understanding of these clouds. We learned that PSCs can form much more rapidly and at higher temperatures than we thought," says Prof. Tom Peter from ETH Zurich.
Scientists from the Max-Planck-Institute for Chemistry, Johannes Gutenberg University Mainz and TU Darmstadt discovered an unexpectedly wide range of particles in their samples. These particles presumably descend from higher stratospheric layers into the polar vortex and contain metals, silicates, black carbon, etc. The origin of these particles is currently being investigated by a group at Mainz as part of the project entitled “In situ experiments on the chemical composition of high-altitude aerosols and clouds in the tropical upper troposphere and lower stratosphere” (EXCATRO), which was awarded an ERC Advanced Grant by the EU.
With regard to another point, the scientists were able to put some of our worries to rest: after the record Arctic winter, there were fears of a dramatic increase in UV radiation in the northern hemisphere. "Although we did find elevated values, they were not nearly as high as would be required to considerably increase the risk of skin cancer," says Marc von Hobe.





























































