Stay Calm with Domino
Preparation of Benzodiazepine-Containing Drugs
Drugs such as sedatives, hypnotics, and anticonvulsants often contain a benzodiazepine moiety. They are useful in the treatment of, for example, anxiety, insomnia, agitation, and seizures. The 1,5-benzodiazepin-2-one skeleton in particular is considered a “privileged” scaffold in medicinal chemistry and drug development. The pharmacological importance of benzodiazepine derivatives therefore justifies new efforts for their synthesis. As recently reported in the European Journal of Organic Chemistry, a Columbian/French team led by Yoann Coquerel and Jean Rodriguez have now introduced a general domino sequence to provide new synthetic entry to 1,3-dihydro-2H-1,5-benzodiazepin-2-ones.
An important focus of modern organic synthesis is economy, and many transformations have been developed to allow the creation of molecular complexity and functional diversity in a single chemical operation. These transformations are best performed with polyfunctionalized substrates in which the distinct reactive groups react selectively in a well-defined order to give a single product. α-Oxo ketenes exhibiting two carbonyl groups and one double bond distributed over three carbon atoms are highly reactive molecules that fall into this class of compounds. α-Oxo ketenes have already been used to prepare a number of heterocycles, and the scientists have already shown that these compounds can be obtained through the rearrangement of 2-diazo-1,3-dicarbonyl compounds. The team was further interested in the reactions of 2-diazoalkane-1,3-diones with bis(N-nucleophiles) in a domino sequence to prepare the desired benzodiazepine derivatives.
Although synthetic routes to benzodiazepines exist, they are often sensitive to the type of substrate and the reaction conditions; moreover, these methods usually require long reaction times and are often complicated by side reactions. Importantly, using their domino methodology and without any additives, the researchers found that microwave irradiation of a 1:1 mixture of a set of diazo compounds with o-phenylenediamine furnished a series of bi- and tricyclic 1,3-dihydro-2H-1,5-benzodiazepin-2-ones, which are among the most promising compounds containing the benzodiazepine structure. The authors postulate that the reaction follows this sequence: rearrangement, nucleophilic addition, and intramolecular imination. This reaction represents a new and advantageously complementary synthetic approach to a valuable class of drug-like compounds.
Original publication
Most read news
Original publication
Yoann Coquerel et al.; "Microwave-Assisted Domino Benzannulation of α-Oxo Ketenes: Preparation of 1,3-Dihydro-2H-1,5-benzodiazepin-2-ones"; European Journal of Organic Chemistry
Topics
Organizations
Other news from the department science

Get the chemical industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.

Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.
Last viewed contents
James_Parker_(cement_maker)

Synthesizing valuable chemicals from contaminated soil - Researchers use electrolysis to produce dichloro and dibromo compounds in a safer and more environmentally friendly manner

Wet electrolyte could be a key to inexpensive energy storage - Water containing battery electrolyte could enable cheaper, easier to produce batteries

New magnetic behavior in nanoparticles could lead to even smaller digital memories
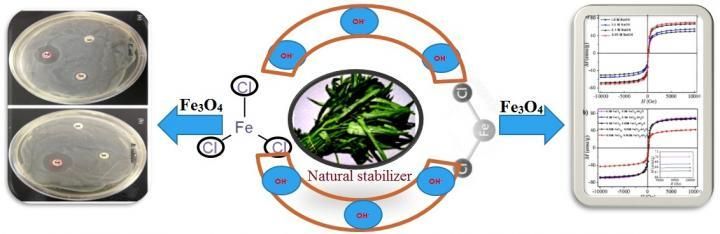
Eco-friendly method for the synthesis of iron oxide nanoparticles - Producing nanomaterials from plant extracts
François_Diederich

One-way roads for spin currents
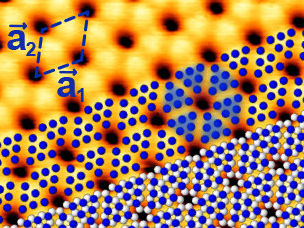
Blue phosphorus - mapped and measured for the first time
Category:Swiss_biochemists
Setting the gold standard
Bayer reduces size of Board of Management from seven to five members