A microscopic look at atmospheric chemistry
New discovery contradicts textbook models
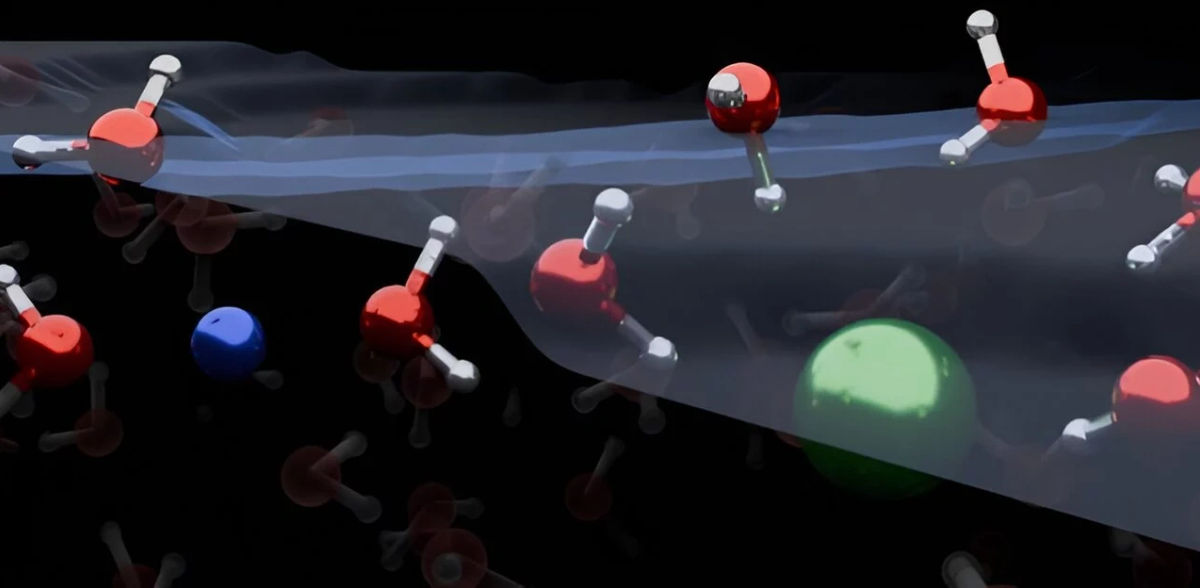
Many important reactions related to climate and environmental processes take place where Water molecules interface with air. For example, the evaporation of ocean water plays an important role in atmospheric chemistry and climate science. Understanding these reactions is crucial to efforts to mitigate the human effect on our planet. Textbook models will now need to be re-drawn after a team of researchers has found that water molecules at the surface of salt water are organized differently than previously thought.
The distribution of ions at the interface of air and water is relevant for many atmospheric processes. However, a precise understanding of the microscopic processes has so far been intensely debated. In a publication published in the journal Nature Chemistry, researchers at the Max Planck Institute for Polymer Research in Germany show that ions and water molecules at the surface of most salt-water solutions, known as electrolyte solutions, are organized in a completely different way than traditionally understood. This could lead to better atmospheric chemistry models and other applications.
The researchers set out to study how water molecules are affected by the distribution of ions at the exact point where air and water meet. Traditionally, this has been done with a technique called vibrational sum-frequency generation (VSFG). With this technique using laser radiation, it is possible to measure molecular vibrations directly at these key interfaces. However, although these measurements can measure the strength of the signals, they do not measure whether the signals are positive or negative, which has made it difficult to interpret findings in the past, as the sign of the signal reflects whether water is up- or down-oriented at the surface. Additionally, using experimental data alone can give ambiguous results.
The team overcame these challenges by utilizing a more sophisticated form of VSFG called heterodyne detected (HD)-VSFG to study different electrolyte solutions. They then developed advanced computer models to simulate the interfaces in different scenarios.
The combined results showed that both positively charged ions, called cations, and negatively charged ions, called anions, are depleted from the water/air interface. The cations and anions of simple electrolytes orient water molecules in both up- and down-orientation. This is a reversal of textbook models, which teach that ions form an electrical double layer and orient water molecules in only one direction.
Co-first author Dr. Yair Litman, who meanwhile holds a DFG Fellowship as a Theoretical Chemist in the Yusuf Hamied Department of Chemistry at the University of Cambridge, said: “Our work demonstrates that the surface of simple electrolyte solutions has a different ion distribution than previously thought and that the ion-enriched subsurface determines how the interface is organized: going from air into the bulk salt solution, one first encounters a few layers of pure water, then comes a layer enriched in ions, before reaching the bulk.”
Co-first author Dr. Kuo-Yang Chiang of the Max Planck Institute said: “This paper shows that combing high-level HD-VSFG with simulations is an invaluable tool that will contribute to the molecular-level understanding of liquid interfaces.”
Prof. Mischa Bonn, who heads the Molecular Spectroscopy deparment of the Max Planck Institute, adds: “These types of interfaces occur everywhere on the planet, so studying them not only helps our fundamental understanding but can also lead to better devices and technologies. We are applying these same methods to study solid/liquid interfaces, which could have potential applications in batteries and energy storage.”
Original publication
Other news from the department science

Get the chemical industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
Most read news
More news from our other portals
Last viewed contents
120 in one sweep - Comprehensive concept optimizes the production of numerous specialty polyesters
New superconducting coil improves MRI performance - Research offers higher resolution, shorter scan time

Electrified Diamonds: Basel Physicists on the Trail of Quantum Information
Cortisporin
Karl_Friedrich_Mohr
Promega and Leica Microsystems Sign Agreement to Develop Fluorescent Ligands Enabling Live Cell Imaging at the Sub 100 nm Range

A candlelight-like glow from a flexible organic LED
Mustard_plant
First-ever videos show how heat moves through materials at the nanoscale and speed of sound