Novel multi-proton carrier complex as efficient proton conductor at high temperatures
Researchers develop a highly symmetric ruthenium (III) complex with six imidazole-imidazolate groups for efficient high-temperature proton conduction in fuel cells
As the world is moving towards more environment-friendly and sustainable sources of energy, fuel cells are receiving a lot of attention. The main advantage of fuel cells is that they use hydrogen, a clean fuel, and produce only water as a by-product while generating electricity. This new and clean source of electricity could replace conventional lithium-ion batteries, which currently power all modern electronic devices.
Researchers develop a highly symmetric ruthenium (III) complex with six imidazole-imidazolate groups for efficient high-temperature proton conduction in fuel cells
Tokyo University of Science
Most fuel cells use a Nafion membrane— a synthetic polymer-based ionic membrane— which serve as a water-based proton conducting solid electrolyte. The use of water as a proton conduction medium, however, creates a major drawback for the fuel cell, namely the inability to function properly at temperatures above 100 ֯ C, the temperature at which water starts to boil, leading to a drop in proton conductivity. Therefore, there is a need for new proton conductors that can transfer protons efficiently even at such high temperatures.
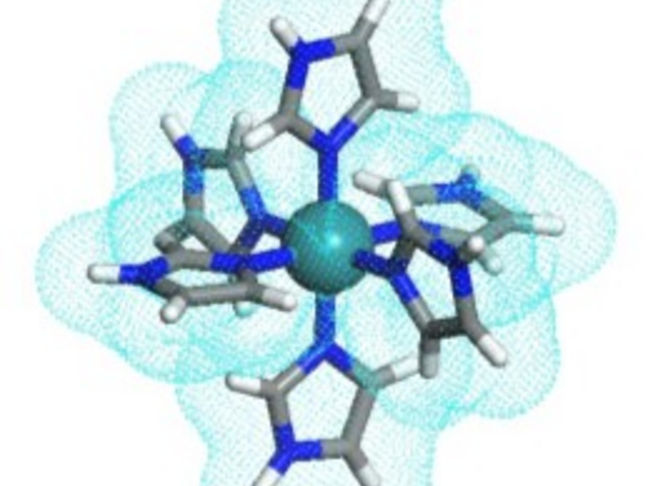
In a recent breakthrough, a team of researchers from Japan, led by Prof. Makoto Tadokoro from Tokyo University of Science (TUS), reported a novel imidazole-imidazolate metal complex based high-temperature proton conductor that shows efficient proton conductivity even at 147°C (Fig.1). The research team included Dr. Fumiya Kobayashi from TUS, Dr. Tomoyuki Akutagawa and Dr. Norihisa Hoshino from Tohoku University, Dr. Hajime Kamebuchi from Nihon University, Dr. Motohiro Mizuno from Kanazawa University, and Dr. Jun Miyazaki from Tokyo Denki University. “Imidazole, a nitrogen-containing organic compound, has gained popularity as an alternative proton conductor for its ability to operate even without water. However, it has a lower proton transfer rate than conventionally used Nafion and melts at 120°C. To overcome these issues, we introduced six imidazole moieties into a ruthenium (III) ion to design a new metal complex that operates as a multi-proton carrier and has high temperature stability,” explains Prof. Tadokoro when asked about the rationale behind their study.
The team designed a new molecule where three imidazole (HIm) and three imidazolate (Im-) groups were attached to a central ruthenium (III) ion (Ru3+). The resulting single molecular crystal was highly symmetrical and resembled a “starburst” shape. Upon investigating the proton conductivity of this starburst-type metal complex, the team found that each of the six imidazole groups attached to the Ru3+ ion acts as a proton transmitter. This made the molecule 6 times more potent than individual HIm molecules, which could only transport one proton at a time.
The team also explored the mechanism underlying the high-temperature proton conduction ability of the starburst molecules. They found that at a temperature of more than –70°C, the proton conductivity resulted from individual localized rotations of the HIm and Im- groups and proton jump to other Ru(III) complexes in the crystal via hydrogen bonds. At temperatures beyond 147°C, however, the proton conductivity arose from whole-molecule rotation, which was also responsible for the superior proton conductivity at high temperatures. This rotation, confirmed by the team using a technique called “solid-state 2H-NMR spectroscopy,” resulted in a conductivity rate three orders of magnitude larger (σ = 3.08 × 10-5 S/cm) than that for individual HIm molecules (σ = 10-8 S/cm).
The team believes that their study could act as a new driving principle for proton-conducting solid-state electrolytes. The insights from their novel molecular design could be used to develop new high-temperature proton conductors as well as improve the functionality of the existing ones. “ Fuel cells hold the key to a cleaner and greener tomorrow. Our study offers a roadmap for improving the performance of these carbon-neutral energy resources at high temperatures by designing and implementing molecular proton conductors that can transfer protons efficiently at such temperatures, ” concludes Prof. Tadokoro.
Hopefully, we’re not too far from realizing a future based on fuel cells and clean energy.




























































