Oxide tuning by ion transfer
Researchers discover new formula for changing the electronic and magnetic properties of oxide interfaces
Most materials are either magnetic, or they are not. However, scientists at Forschungszentrum Jülich have now discovered a new mechanism that allows for tailoring the electronic and magnetic properties of a material in a targeted and reversible manner. The effect depends on the transfer of ions at the interface of two oxides - the researchers were able to prove the existence of this process for the first time experimentally. Both oxides alone typically show neither magnetism nor significant electrical conductivity. Only in combination, both properties occur at the interface. The quantitative relationships are still unclear. However, the researchers succeeded in changing the magnetic order at the interface by moving ions across the interface, making the material more magnetic. They were also able to control the electrical properties via the resulting electron distribution.
Material systems with such flexible electronic properties are relevant for various emerging IT technologies such as neuromorphic computing or spintronic approaches. One potential application would be a "multifunctional" transistor that can control not only electric current but also spin currents (spintronics). Such a device could then be controlled by applying both a voltage and a magnetic field.
The "electronic" interface system consisting of the oxides LaAlO3 and SrTiO3 was already discovered in 2004 and has caused worldwide interest. Both materials exchange both electrons and atomic components in the form of charged cations at the interface, as the Jülich researchers have now been able to show. New electronic properties are thus formed at the interface. A similar phenomenon is also known from semiconductors. However, the purely electronic effect is limited exclusively to the transfer of electrons.
Researchers at the Jülich Peter Grünberg Institute (PGI-7) have now been able to demonstrate experimentally for the first time that, in addition to the exchange of electrons, "ionic" charge transfer is also responsible for the change in the electronic properties of the LaAlO3/SrTiO3 system. The concept allows for adjusting the conductivity and magnetic properties of the interface at the same time.
By means of various "mixtures" of electronic and ionic charge transfer, they were able to create different interfaces that differ in terms of their electronic properties and atomic structure. For example, the researchers were able to create interfaces with high conductivity and weak magnetism or with lower conductivity and stronger magnetism.
Experimental insights were obtained by using X-ray photoelectron spectroscopy under atmospheric conditions (near-ambient pressure X-ray photoelectron spectroscopy, NAP-XPS). The method is still quite new and offers - as has now been demonstrated - direct access to the ionic processes at interfaces with atomic precision. Experimentally, the movement of cations across the interface can be studied and dynamically controlled by temperature and oxygen atmosphere. From these data, specific conclusions can be drawn about the relationship between ionic structure and the resulting electric and magnetic properties.
Process control by oxygen contact
The ion transfer is controlled by contact with oxygen. This causes strontium ions (Sr) to move across the interface. Each missing Sr ion binds two electrons. Because these can no longer contribute to electrical conductivity, the electrical conductivity decreases. At the same time, this process causes crystal defects which influence the magnetic order of the remaining electrons. Thus the system becomes more magnetic while losing conductivity. The fact that these cations can move freely to this extent was previously postulated by researchers, but most of them did not consider it possible in practice. Therefore, the present study reflects a milestone in understanding ionic processes at oxide interfaces.
Original publication
Original publication
M. Rose, B. Šmíd, M. Vorokhta, I. Slipukhina, M. Andrä, H. Bluhm, T. Duchoň, M. Ležaić, S. A. Chambers, R. Dittmann, D. N. Mueller*, F. Gunkel*; "Identifying Ionic and Electronic Charge Transfer at Oxide Heterointerfaces"; Advanced Materials 2004132 (published online 2 December 2020)
Organizations
Other news from the department science

Get the chemical industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
Most read news
More news from our other portals
Last viewed contents
Category:Ribonucleases
Croda_International
Caliper Creates Specialized ACES Team to Deliver Customized Technology Solutions - Initiative to Deliver Best-In-Class Solutions for Laboratory Automation
Cardenolide
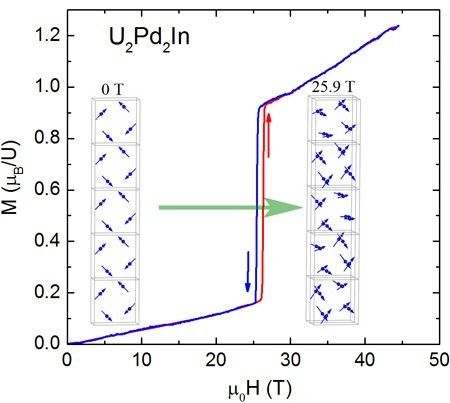
Not everything is ferromagnetic in high magnetic fields
DSM completes acquisition of PTG
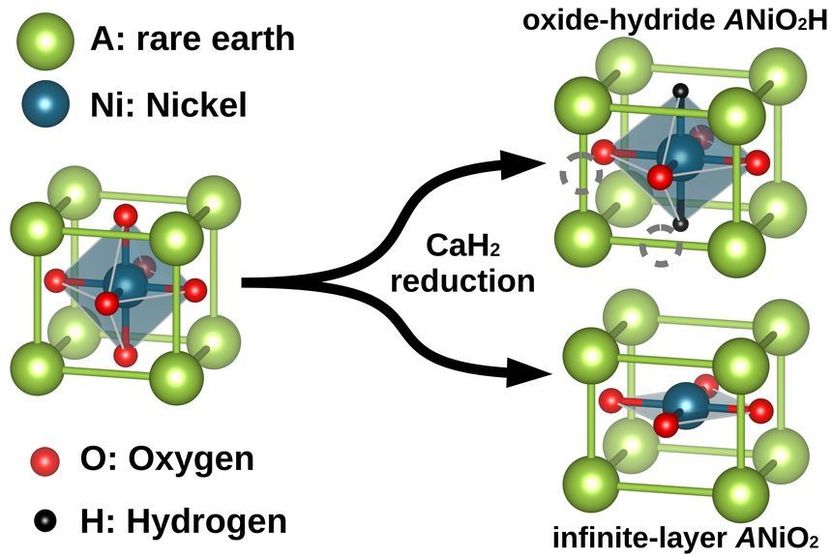
Superconductivity: It’s Hydrogen’s Fault - With new nickelates and the predictive power of supercomputers to the superconductor without any cooling

New class of stable nickel complexes developed - Easy-to-handle nickel complexes for practical application in nickel catalysis