When crystals throw sparks
Voltage can be used for chemical reactions: New model for pyroelectric reactions developed
pyroelectricity is a phenomenon in physics in which heat can be converted into electricity via certain Crystals or into a voltage that can be used for chemical reactions. While the first application is already used in devices such as motion detectors today, the second application, although well-known, has not yet been sufficiently described on a theoretical basis. A team of physicists at TU Bergakademie Freiberg has now found a way to describe and predict the processes on pyroelectric surfaces.

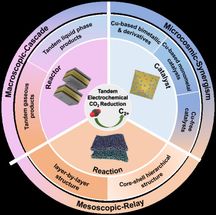
Photograph of the lithium tantalate crystal with flashovers.
TU Bergakademie Freiberg / Sven Jachalke
Dr. Mateo de Vivanco picks up a hair dryer and directs warm air onto an inconspicuous small crystal. A moment later, after about 5 seconds, small flashes are visible to the naked eye on the surface of the crystal (see photo). What sounds like magic can be described with the phenomenon of pyroelectricity, which is sufficiently well known in physics: "Crystals of the pyroelectric material class generate an electrical voltage when their temperature changes," explains the research assistant at the Institute for Experimental Physics (IEP) of the TU Bergakademie Freiberg. The reason for this phenomenon lies in the smallest positively and negatively charged particles of the crystal, which drift together or apart once the temperature changes. These movements sum up to an electric discharge current at the surface of the crystal.
Voltage can be used for chemical reactions
In a possible application scenario the resulting voltage could be used for chemical reactions. "Of particular interest is the splitting of water into oxygen and hydrogen, which is a gas in great demand as an energy carrier and in the chemical industry," says Dr. Mateo de Vivanco. However, although this reaction could be experimentally demonstrated several years ago, the physico-chemical processes running behind have so far been insufficiently described in international research. This is where the team of physicists came in and, as a first step, studied existing models that explain similar reactions.
"As a chemist in a physics research group, I wanted to calculate the hydrogen yield from pyroelectric water splitting. Since this was not possible with existing models, my team and I considered which factors limit the pyroelectric yield," explains the corresponding author of the study. "Compared to the direct use of (pyro-)electricity, a chemical reaction such as hydrogen production involves overvoltages of various kinds, which can impede water splitting," Dr. Mateo de Vivanco continues. After several years of research, the team thus succeeded in developing the chemical-physical model that has now been presented, which can be used to explain and predict the processes on pyroelectric surfaces in chemically labile media such as water.
Model enables understanding of the complex process
Thanks to the new model, which the researchers present in the August issue of the journal "Physical Chemistry Chemical Physics", new possibilities for understanding complex electrochemical processes on solid surfaces are opening up. For the first time, the amount of hydrogen produced can be explained and predicted. If the pyroelectric process is further developed in the future - not only with the help of the new model - new potential applications for research and industry will arise.
"The team's current results represent a reliable lead-up to the current discussions, in particular when it comes to the development of essential hydrogen-based technologies", says Prof. Dirk C. Meyer, Director of the IEP, in his capacity as Scientific Spokesman of the Center for Efficient High-Temperature Materials Conversion (ZeHS) at TU Bergakademie Freiberg. "The study of crystal-physical coupling phenomena, especially pyroelectricity, has been a focal point of the work at the IEP for several years now", says Prof. Dirk Meyer.
Original publication
Most read news
Original publication
Mateo U. de Vivanco, Matthias Zschornak, Hartmut Stöcker, Sven Jachalke, Erik Mehner, Tilmann Leisegang & Dirk C. Meyer; "Pyroelectrically-driven chemical reactions described by a novel thermodynamic cycle"; Physical Chemistry Chemical Physics; 2020
Organizations
Other news from the department science

Get the chemical industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
Most read news
More news from our other portals
Last viewed contents
Norvatis: New study shows Elidel® provides sustained control of eczema

From scrap to raw material: With artificial intelligence to an efficient circular economy - Car2Car project develops technologies for an optimized recycling of end-of-life vehicles
Good vibrations help reveal molecular details

Physicists propose a new method for monitoring nuclear waste - Scenarios for using neutrino detectors in nuclear interim storage facilities

Brenntag acquires North American acetone specialist Matrix Chemical
SABIC selects Basell's Hostalen technology for two new HDPE plants in Europe and Saudi Arabia
Affymetrix and Iconix Collaborate to Develop New Solutions for Assessing Drug Toxicity

Electrons zipping around in crystals

Lignovations Closes Seed Investment to Accelerate Lignin-Based Biomaterial Innovation - A multifunctional alternative to many synthetic chemicals in cosmetics, coatings, packaging, adhesives, and other applications
Breaking down plastic waste - Organo-ruthenium mediated hydrogenation under mild conditions
AkzoNobel to outline new strategy - Plan for enhanced value creation