Two-Dimensional Carbon Networks
Graphdiyne as a Functional Lithium-Ion Storage Material
lithium-ion batteries usually contain graphitic carbons as anode materials. Scientists have investigated the carbonic nanoweb graphdiyne as a novel two-dimensional carbon network for its suitability in battery applications. Graphdiyne is as flat and thin as graphene, which is the one-atomic-layer-thin version of graphite, but it has a higher porosity and adjustable electronic properties. In the journal Angewandte Chemie, researchers describe its simple bottom-up synthesis from tailor-made precursor molecules.
© Wiley-VCH
Carbon materials are the most common anode materials in lithium-ion batteries. Their layered structure allows lithium ions to travel in and out of the spaces between layers during battery cycling, they have a highly conductive two-dimensional hexagonal crystal lattice, and they form a stable, porous network for efficient electrolyte penetration. However, the fine-tuning of the structural and electrochemical properties is difficult as these carbon materials are mostly prepared from polymeric carbon matter in a top-down synthesis.
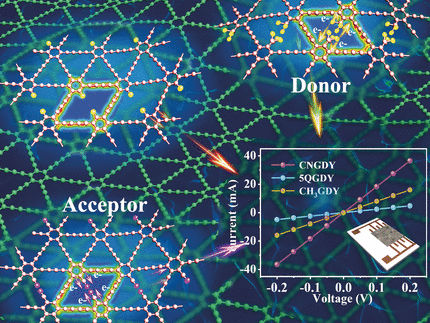
Graphdiyne is a hybrid two-dimensional network made of hexagonal carbon rings bridged by two acetylene units (the “diyne” in the name). Graphdiyne has been suggested as a nanoweb membrane for the separation of isotopes or helium. However, its distinct electronic properties and web-like structure also make graphdiyne suitable for electrochemical applications. Changshui Huang from the Chinese Academy of Sciences, Beijing, and colleagues have investigated the lithium-storage capabilities and electrochemical properties of tailor-made, electronically adjusted graphdiyne derivatives.
The scientists synthesized the graphdiyne derivatives in a bottom-up strategy by adding precursor molecules on a copper foil, which self-organized to form ordered layered nanostructures. Using monomers containing functional groups with interesting electronic properties, the authors prepared functional graphdiynes with distinct electrochemical and morphological properties.
Among these functional groups, those exerting electron-withdrawing effects reduced the band gap of graphdiyne and increased its conductivity, the authors reported. The cyano group was especially effective and, when used as an anodic material, the cyano-modified graphdiyne demonstrated excellent lithium-storage capacity and was stable for thousands of cycles, as the authors reported.
In contrast, when graphdiyne was modified with bulky functional groups (methyl groups) that donate electrons to the graphdiyne network, the authors observed a larger layer spacing, which made the material structure unstable so that the anode only survived a few charge and discharge cycles. The authors also compared both modified graphdiyne materials to an “empty” version where only hydrogen occupied the position of the functional groups in the network.
The authors conclude that modified graphdiyne can be prepared by a bottom-up strategy, which is also best suited to build functional two-dimensional carbon material architectures for batteries, capacitors, and other electrocatalytic devices.
Original publication
Other news from the department science
These products might interest you

Battery Testing Services by Battery Dynamics
Learn more about the performance and service life of your battery cells in less time
Benefit from modern measurement technology and an experienced team

Batt-TDS by ystral
YSTRAL Batt-TDS mixing and dispersing machine
Boost your battery slurry process

Get the chemical industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
Most read news
More news from our other portals
See the theme worlds for related content
Topic World Battery Technology
The topic world Battery Technology combines relevant knowledge in a unique way. Here you will find everything about suppliers and their products, webinars, white papers, catalogs and brochures.

Topic World Battery Technology
The topic world Battery Technology combines relevant knowledge in a unique way. Here you will find everything about suppliers and their products, webinars, white papers, catalogs and brochures.
Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.

Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.
Last viewed contents

Cloud Formation: How Feldspar Acts as Ice Nucleus
GlaxoSmithKline
Category:Calcium_channel_blockers
Amsterdam_Diamond
Quick and reliable 3-D imaging of curvilinear nanostructures