The modern gold rush
Advertisement
Chemists in the UK have sythesised stable cationic gold(I) alkene complexes using a bulky phosphine ligand and strained alkenes. Alkenes are major products of the petrochemical industry and the way they interact with metals often dictates their chemistry. There has been extensive research carried out on the interaction of alkenes with mid transition metals, however only recently has gold chemistry gained momentum, with many researchers describing it as the modern gold rush.
Indeed, recent gold-alkene chemistry includes work by Widenhoefer and colleagues, who synthesised two coordinate gold(I) π-alkene complexes, and characterised them by X-ray diffraction.
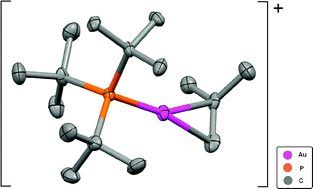
Now, Christopher Russell, Mike Green and colleagues at the University of Bristol, and John McGrady at the University of Glasgow, have studied the interaction of alkenes with a gold-phosphine cationic centre. They show that the use of the sterically demanding and strong σ-donor phosphine ligand (PtBu3) allows the formation of remarkably stable complexes using strained alkenes such as norbornene, norbornadiene, trans-cyclooctene and isobutylene.
Russell and his team wanted to explore whether the use of strained alkenes would stabilise the metal-alkene bond, in order to allow them to probe the physical and electronic structure of the bond. They found that the catalytically important gold-alkene cationic systems were indeed stable giving insights into the structure and mechanisms involved.
‘Our work has just scratched the surface of this important interaction, but our observations have implications for the reactivity of alkenes at gold centres,’ says Russell. ‘The applications of the newly discovered chemistry of gold has obvious implications in the field of homogeneous catalysis, however there may be insights which also apply to the burgeoning fields of heterogeneous catalysis and nanotechnology,’ he adds.
Original publication: Green et. al., Chem. Commun., 2009.
































































